
compared to solids and liquids, gases are easily compressed - gases expand to fill the volume of their containers molecules of different gases readily mix with each other the real-life behavior of all gases is identical of that of ideal gases gases expand to fill the volume of their containers 
Pressure and temperature identify the correct statements about the behavior of gases Straight line For an ideal gas, predict the shape of the plot for each of these graphs, where P is pressure, V is volume, and T is absolute temperature Pressure and 1/V graph with P on y-axis and 1/V on x-axis Pressure and volume (P and V) For an ideal gas, predict the shape of the plot for each of these graphs, where P is pressure, V is volume, and T is absolute temperature P and V For an ideal gas, predict the shape of the plot for each of these graphs, where P is pressure, V is volume, and T is absolute temperature Which line of the graph corresponds to the dataġ.7 Which are directly proportional and which are inversely proportional? the same number of choco pies were sold each year - the percent increase in sales of choco pies was approx the sam e in each year study the experimental data and graph the percent increase in sales of choco pies was approx the sam e in each year the rate of sales of choco pie was greater in 2000 the rate of sales of choco pie was greater in 2010 Sam is curious about the sales of choco pies, and he wants to see if other people like them as much as he does.Ĭonsider the two graphs showing sales data for choco pies in 20 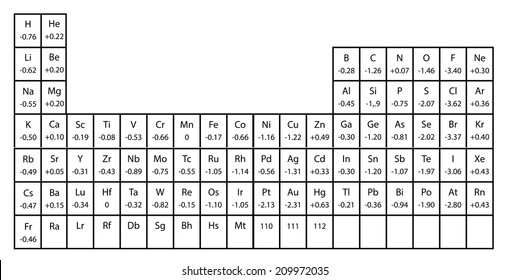
He finds them very tasty and can eat boxes of choco pies at a time. How many times did the student flip the coin? 100 Sam recently discovered a new type of snack, choco pie. He then created a pie chart from the data. M Line graph of the progress of a person on a long hike is shown in the graphīetween which points was the hiker movingīetween which points is the hiker stationary:ī&C A student flipped a coin many times and recorded the frequency of heads and tails. The y-axis should be labeled _ diameterĬircumference use the graph to determine the units of the slope and y-intercept Regular Italian dressing Diameter (cm) //Circumference (cm) Type of dressing and if it dissolves in water She recorded the results in her notebook. In her experiment, she tested which products were completely soluble in water. She knows that lipids do not dissolve in water. when the instructor is conducting a demonstration with chemicals all of them Marissa was trying to determine if certain products contained lipids (fats) or not. when a classmate in the lab is working with chemicals when the student is working with chemicals Estimate the volume to the nearest 5 mL increment and enter it with the proper number of significant figures 200 mL Identify the situation which require a student to wear goggles (117 km/h)- Read the volume of the liquid in the beaker. Electronegativity increases while moving across a period and decreases down a group.Read the speedometer and report the speed to the proper number of digits in miles per hour (mph) and kilometers per hour (km/h).Their electron affinity is high and they also have high ionization values.Thus, they gain one or two electrons to fulfil their octet.These elements like F, Cl, and S have one or two electrons short of achieving their octet in the outermost shell.They have a high tendency of attraction toward electrons in a bond.Thus, the order of decreasing electropositivity is Na > Mg > Al. So, the electropositivity will also decrease as we move from Sodium to magnesium to Aluminium.As we move from left to right along a period, we know that the electropositivity decreases.Magnesium (Mg), Sodium (Na), Aluminium (Al) all are metals and belong to the same period 3 of the periodic table.The elements at the left side and bottom of the periodic table are the most electropositive in nature.The increase in the size of atoms increases the electropositivity of the elements as we move down the group.As we move from left to right across a period, the atomic radius decreases as a result of which the electronegativity increases and the electropositivity decreases.The energy required to take out an electron from its outer shell is also low. 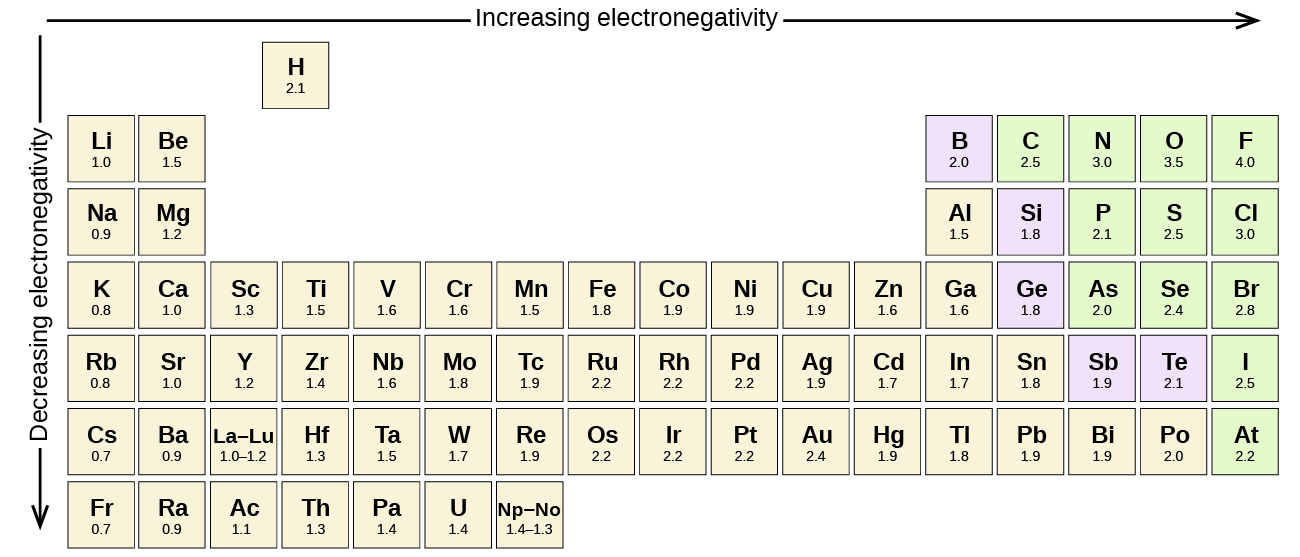
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
